Research
Welcome to the Weiberg Lab: Pathogen small RNAs as host immunity suppressors
Please, visit us also on www.weiberglab.org
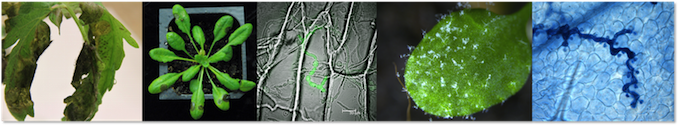
Our team is investigating how plant pathogens use small RNAs to support infection of their plant hosts.
On the one hand, we are studying Botrytis cinerea, an aggressive plant fungal pathogen that can cause necrotic lesions known as the Botrytis Gray Mold Disease on hundreds of different host plant species. On the other hand, we use Hyaloperonospora arabidopsidis, which is a highly adapted obligate biotrophic oomycete pathogen that exclusively infects Arabidopsis thaliana, causing the Downy Mildew Disease.
Cross-Kingdom RNA Interference
Botrytis and Hyaloperonospora release small RNA effectors during host colonization that enter plant cells and hijack the plant Argonaute (AGO)/RNA-induced gene silencing complex (RISC) to induce host gene silencing for infection. This infection strategy is called cross-kingdom RNA interference (RNAi). We are using Botrytis and Hyaloperonopsora pathogens to elucidate the molecular mechanisms and functions of cross-kingdom RNAi in fungal and oomycete pathogens.
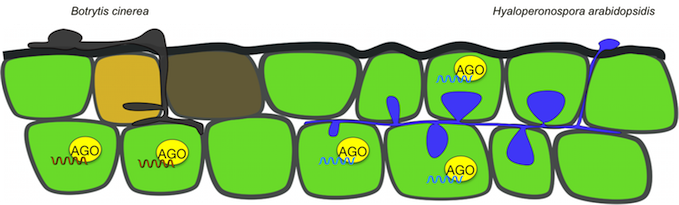
We employ a diverse set of genomics, transcriptomics, bioinformatics, molecular genetics, and biochemical approaches to gain fundamental knowledge of the underlying mechanisms of cross-kingdom RNAi. Understanding these molecular mechanisms and factors that determine cross-kingdom RNAi bears enormous potential in its application for RNA-based biotechnology approaches.
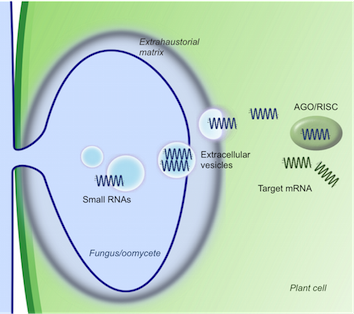
Extracellular Vesicles
A fundamental question of the cross-kingdom RNAi phenomenon is the transport of small RNAs between filamentous pathogens and their host plants. Extracellular vesicles (EVs) have become famous to mediate cell-to-cell communication in mammals; with small RNAs are important cargoes in EV function. We have established protocols to study EV RNAs and are investigating the role of pathogen EVs and their cargoes in the context of plant-microbe interactions.
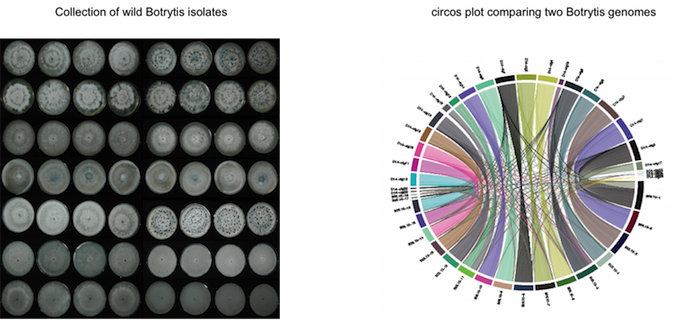
Molecular Diversity of small RNA Effectors
Since small RNA effectors seem to be a common theme among pathogenic microbes, the question arises what is the origin of small RNA effectors and whether there is any selective pressure on the sequence level of small RNA effectors. We are taking comparative genomics and transcriptomics approaches combining Illumina and Nanaopre technologies to study small RNA sequence populations in wild Botrytis field isolates.
Our Research is supported by:


Events:
May 2021, RU5116 EV workshop (postponed)
30th – 31st August 2018, LMU, Mini-Symposium on Extracellular Vesicles in Inter-Organismal Communications



